Apparel & Fashion
Loading subcategories...
View More
Description
COLORCOAT MB4S represents advanced coating technology specifically engineered for pharmaceutical applications requiring superior moisture protection. This ready-to-use polymer blen...
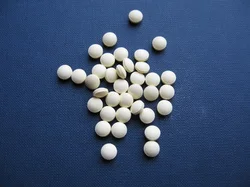
COLORCOAT MB4S represents advanced coating technology specifically engineered for pharmaceutical applications requiring superior moisture protection. This ready-to-use polymer blend combines ethyl cellulose, hydroxypropyl methyl cellulose (HPMC), and polyvinyl alcohol (PVA) to create an effective barrier system for solvent-based coating processes. The formulation delivers excellent film-forming properties that ensure uniform coverage and consistent performance across production batches. Its unique swellable and permeable nature maintains functionality across varying pH levels, making it suitable for diverse drug delivery systems. The pre-formulated nature eliminates mixing errors while reducing preparation time, providing manufacturers with reliable batch-to-batch consistency that meets stringent quality requirements. The pharmaceutical industry relies on COLORCOAT MB4S for moisture-sensitive formulations where product stability determines commercial success. Tablet manufacturers depend on this coating to protect active ingredients from humidity-induced degradation, particularly in tropical climates and during international shipping. Nutraceutical companies utilize it for dietary supplements requiring extended shelf life without refrigeration constraints. Veterinary pharmaceutical producers value its moisture resistance for animal health products stored in challenging environmental conditions. The coating's adaptability supports both immediate-release and modified-release formulations, serving applications ranging from common analgesics to specialized cardiovascular medications with equal effectiveness. Businesses implementing COLORCOAT MB4S gain significant operational advantages through reduced product returns and minimized waste from moisture-related damage. The coating's reliability translates to fewer batch failures and consistent compliance with international regulatory standards. Its technical performance ensures drug efficacy while supporting efficient manufacturing processes. The ready-to-use formulation reduces labor costs and training requirements while maintaining precise formulation consistency. Manufacturers benefit from reduced equipment cleaning time between batches compared to traditional coating systems, resulting in higher production throughput and improved overall manufacturing efficiency without compromising quality standards. Key Features: - Triple polymer blend of ethyl cellulose, HPMC, and PVA for enhanced barrier performance - Ready-to-use formulation requiring no additional preparation or mixing steps - pH-independent swellable and permeable film properties for broad compatibility - Consistent film formation ensuring uniform coverage and protection - Solvent-system compatibility with standard pharmaceutical coating equipment Benefits: - Extended shelf life through effective moisture barrier protection - Reduced production time with ready-to-use formulation requiring no mixing - Broad compatibility with various active ingredients and drug formulations - Consistent batch-to-batch performance meeting quality standards - Efficient manufacturing process with minimal equipment cleaning requirements
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
COLORCOAT MB4S provides robust moisture protection for pharmaceutical solid dosage forms, ensuring uniform coating application that prevents degradation and extends shelf life. Its solvent-based system maintains consistent barrier properties without compromising dissolution rates, making it ideal for sensitive medications in varying environmental conditions. This versatile polymer works with diverse drug formulations while meeting pharmacopeial standards for quality and performance. Manufacturers benefit from the coating's adaptability to existing equipment and consistent performance across production batches. The formulation proves particularly valuable in high-humidity environments where moisture protection is critical for product stability. Its reliable performance supports efficient manufacturing processes while maintaining drug efficacy throughout distribution and storage cycles. - Protective coating for pharmaceutical tablets and capsules against environmental moisture - Barrier protection for hygroscopic drugs during storage and transportation - Enteric coating applications requiring pH-specific drug release profiles - Stability enhancement for nutraceutical pellets and dietary supplements - Moisture-resistant film coating for veterinary medicines with taste-masking properties
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Corel Pharma Chem, Gujarat
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Moisture Barrier Coating Polymer For Solvent Systems
COLORCOAT MB4S moisture barrier coating polymer protects tablets and pills with reliable ethyl cellulose, HPMC, and PVA blend for solvent systems across all pH levels.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Corel Pharma Chem
Manufacturer