Apparel & Fashion
Loading subcategories...
View More
Description
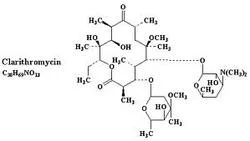
Clarithromycin IP/USP is a semi-synthetic macrolide antibiotic known for its acid stability and enhanced bioavailability compared to earlier macrolides. It works by binding to the ...
Clarithromycin IP/USP is a semi-synthetic macrolide antibiotic known for its acid stability and enhanced bioavailability compared to earlier macrolides. It works by binding to the 50S ribosomal subunit of susceptible bacteria, inhibiting protein synthesis and preventing bacterial growth. This mechanism makes it highly effective against a broad spectrum of Gram-positive and Gram-negative bacteria, including atypical pathogens like Chlamydophila pneumoniae. The product is manufactured under strict quality controls to meet international pharmacopeial standards (IP and USP), ensuring consistency, purity, and reliability for pharmaceutical manufacturers, distributors, and wholesalers. Its chemical stability allows for versatile formulation into tablets, capsules, and oral suspensions, making it a cornerstone ingredient in anti-infective therapy. This antibiotic is extensively used across the pharmaceutical industry for producing medications targeting respiratory infections, skin structure infections, and certain sexually transmitted diseases. Key sectors include generic drug manufacturing, hospital supply chains, and export-oriented pharmaceutical companies requiring WHO-GMP compliant ingredients. It is also integral to veterinary pharmaceuticals for treating bacterial infections in animals. Industries value Clarithromycin for its proven efficacy in treating conditions like acute maxillary sinusitis, pharyngitis, and chronic bronchitis exacerbations, often where penicillin alternatives are needed due to allergy or resistance. Businesses choosing Clarithromycin IP/USP benefit from a reliable supply of high-quality API that reduces production risks and ensures end-product efficacy. Its superior acid stability minimizes side effects like nausea, enhancing patient tolerance and adherence. The product's versatility in formulation and consistent performance helps manufacturers maintain competitive advantage, reduce time-to-market, and build trust with healthcare providers and end-users. Strict quality control processes and compliance with international standards provide assurance of product reliability and therapeutic effectiveness. Key Features: - Complies with IP and USP standards for quality and safety - Broad-spectrum activity against Gram-positive and Gram-negative bacteria - Enhanced acid stability compared to erythromycin - Semi-synthetic macrolide with improved bioavailability - Manufactured under strict quality control standards Benefits: - Reliable therapeutic outcomes for respiratory and skin infections - Reduced gastrointestinal side effects compared to other macrolides - Consistent quality ensures manufacturing process reliability - Versatile formulation options including tablets and oral suspensions - Trusted by pharmaceutical manufacturers worldwide
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Clarithromycin IP/USP is widely used in pharmaceutical formulations for treating bacterial infections affecting the respiratory tract and skin. Its broad-spectrum efficacy makes it a first-line choice for pneumonia, bronchitis, sinusitis, and tonsillitis, especially in cases involving atypical pathogens. This antibiotic is essential for manufacturers producing tablets, capsules, and oral suspensions aimed at both outpatient and clinical settings. Its consistent quality ensures reliable therapeutic outcomes, making it a preferred active pharmaceutical ingredient for serious buyers globally. Industries relying on Clarithromycin include pharmaceutical manufacturing, contract production, and generic drug development. It is also supplied to hospitals, compounding pharmacies, and healthcare distributors who require bulk quantities for formulation. The product's compliance with IP and USP standards guarantees suitability for regulated markets, ensuring consistent performance across various therapeutic applications and manufacturing processes. - Treatment of community-acquired pneumonia and respiratory tract infections in adult and pediatric patients - Management of skin and soft tissue infections caused by susceptible strains of bacteria - Used in combination therapies for Helicobacter pylori eradication in peptic ulcer disease - Prevention and treatment of mycobacterial infections in immunocompromised patients - Formulation into extended-release tablets and oral suspensions for improved patient compliance
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Mehta Pharmaceutical Industries, Maharashtra
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Clarithromycin IP/USP Antibiotic For Respiratory And Skin Infections
Clarithromycin IP/USP is a premium macrolide antibiotic for treating respiratory infections, skin infections, and pneumonia. Trusted by pharmaceutical manufacturers for its reliability and consistent quality.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Mehta Pharmaceutical Industries
Manufacturer