Apparel & Fashion
Loading subcategories...
View More
Description
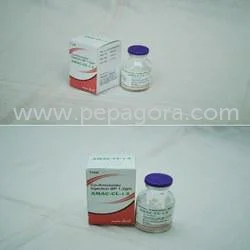
Ceftriaxone Sodium USP 1gm represents pharmaceutical-grade excellence as a third-generation cephalosporin antibiotic active ingredient manufactured to meet stringent United States ...
Ceftriaxone Sodium USP 1gm represents pharmaceutical-grade excellence as a third-generation cephalosporin antibiotic active ingredient manufactured to meet stringent United States Pharmacopeia standards. This high-purity API ensures reliability and effectiveness for injectable formulations, characterized by its broad-spectrum efficacy against both gram-positive and gram-negative bacteria. Supplied as a sterile powder, it maintains optimal stability and integrates seamlessly into various pharmaceutical preparations, providing manufacturers with a trusted component for critical healthcare solutions. This antibiotic API serves multiple sectors within the pharmaceutical industry, particularly in manufacturing injectable antibiotics, generic drug production, and hospital formulations. It supports compounding pharmacies, clinical settings, and global export markets by providing a critical component for infection control and treatment protocols. Industries worldwide rely on its consistent quality for producing medications that address severe bacterial infections while ensuring compliance with international regulatory standards and supporting healthcare providers in delivering effective patient care. For pharmaceutical manufacturers and suppliers, Ceftriaxone Sodium USP 1gm delivers exceptional value through consistent batch-to-batch quality, comprehensive regulatory compliance, and reliable supply chain performance. It minimizes production risks and enhances operational trust, making it ideal for large-volume procurement and specialized market requirements. This API supports cost-efficient manufacturing while maintaining high therapeutic efficacy, helping companies meet global demand for reliable, life-saving antibiotics with confidence and operational dependability. Key Features: - Meets USP grade purity standards for guaranteed quality - 1gm per vial precision dosing for accurate formulation - Broad-spectrum antibacterial activity against diverse pathogens - Sterile powder form optimized for injection preparations - Consistent batch-to-batch reliability and stability Benefits: - Ensures regulatory compliance for global market access - Supports precise dosing in pharmaceutical manufacturing - Provides effective treatment against multiple bacterial types - Maintains product integrity through sterile formulation - Delivers reliable performance for critical healthcare applications
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Ceftriaxone Sodium USP 1gm is a broad-spectrum cephalosporin antibiotic active ingredient essential for manufacturing injectable medications. This high-purity API ensures consistent quality and compliance with global regulatory standards, making it ideal for pharmaceutical production facilities requiring reliable components for critical healthcare solutions. Its sterile powder formulation guarantees stability and seamless integration into various injectable preparations, supporting both large-scale manufacturing and specialized medical formulations. Trusted by pharmaceutical manufacturers and healthcare suppliers worldwide, this product delivers dependable performance for infection control treatments. The API's rigorous quality control and batch-to-batch consistency make it suitable for global distribution networks, meeting the demands of importers and wholesalers who require pharmaceutical-grade ingredients for life-saving medications and therapeutic applications. - Manufacturing injectable antibiotics for bacterial meningitis treatment - Production of medications for severe respiratory infections - Formulating solutions for skin and soft tissue infection management - Creating urinary tract infection treatments and surgical prophylaxis - Developing septicemia control medications and hospital infection protocols
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Mac Heal Laboratories Ltd, Gujarat
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Ceftriaxone Sodium USP 1gm API For Injectable Antibiotic Production
Ceftriaxone Sodium USP 1gm is a high-purity antibiotic API for injectable pharmaceutical production, meeting strict USP standards for reliable treatment of severe bacterial infections in clinical and manufacturing applications.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Mac Heal Laboratories Ltd
Health & Personal CareManufacturer