Apparel & Fashion
Loading subcategories...
View More
Description
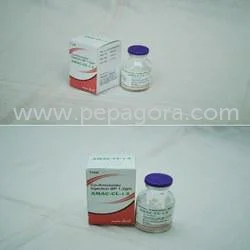
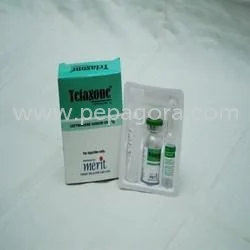
Ceftazidime Injection USP 1gm represents a sterile antibiotic formulation containing Ceftazidime Pentahydrate equivalent to 1000mg Ceftazidime, combined with Sodium Carbonate for o...
Ceftazidime Injection USP 1gm represents a sterile antibiotic formulation containing Ceftazidime Pentahydrate equivalent to 1000mg Ceftazidime, combined with Sodium Carbonate for optimal stability and solubility. As a third-generation cephalosporin, it demonstrates exceptional broad-spectrum activity against challenging Gram-negative pathogens including Pseudomonas aeruginosa strains. Manufactured under rigorous aseptic conditions that comply with United States Pharmacopeia standards, this injection guarantees purity, consistent potency, and patient safety. Designed for either intravenous or intramuscular administration, it delivers rapid therapeutic action precisely when prompt antibiotic intervention becomes critical for patient outcomes. The product packaging maintains sterility throughout storage and handling while facilitating straightforward administration for healthcare professionals. This injection serves as fundamental treatment across hospital intensive care units, surgical centers, emergency departments, and specialized clinics managing serious infections. Medical professionals utilize it extensively in oncology departments for neutropenic patients, burn units for sepsis prevention, and critical care for ventilator-associated pneumonia cases. Pharmaceutical distributors supply this product to government healthcare programs, private hospital networks, and emergency medical services, ensuring continuous availability for life-saving interventions. Its proven reliability establishes it as a cornerstone in infectious disease management protocols within modern healthcare systems worldwide, supporting consistent patient care standards. For importers, distributors, and healthcare suppliers, this product delivers substantial business value through its unwavering quality, regulatory compliance, and sustained market demand. Its reliability minimizes risks associated with product efficacy concerns or supply chain disruptions, building enduring trust with medical facilities and practitioners. The product's consistent performance and therapeutic effectiveness create repeated procurement cycles and long-term partnership opportunities within competitive pharmaceutical distribution networks. By incorporating this critical-care injection into their portfolio, businesses secure a proven product with steady demand that strengthens their market position and supports reliable revenue streams. Key Features: - Contains Ceftazidime Pentahydrate equivalent to 1000mg Ceftazidime per vial - Sterile formulation with Sodium Carbonate ensuring stability and solubility - Manufactured under strict aseptic conditions meeting USP standards - Effective against Gram-negative bacteria including Pseudomonas aeruginosa - Suitable for both intravenous and intramuscular administration methods Benefits: - Delivers reliable broad-spectrum coverage for severe bacterial infections - Ensures patient safety through sterile manufacturing and quality control - Provides flexible administration options for different clinical scenarios - Supports healthcare professionals with consistent therapeutic performance - Meets stringent regulatory standards for pharmaceutical distribution
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Ceftazidime Injection USP 1gm delivers potent antibacterial action against serious infections where other treatments fail. This third-generation cephalosporin targets Gram-negative bacteria including Pseudomonas aeruginosa, providing critical care for hospitalized patients through intravenous or intramuscular administration under medical supervision. Its sterile formulation ensures consistent safety and therapeutic effectiveness in diverse clinical environments. Healthcare facilities rely on this injection for managing complex infections across multiple departments. It serves intensive care units, surgical wards, emergency rooms, and outpatient clinics with proven results. Medical distributors and wholesalers value its compliance with USP standards and consistent performance, making it essential inventory for meeting healthcare provider demands and patient treatment protocols. - Managing severe pneumonia and lower respiratory tract infections in critical care settings - Treating complicated urinary tract infections including pyelonephritis cases - Addressing intra-abdominal infections such as peritonitis and appendicitis - Combating bacterial septicemia and bloodstream infections in hospitalized patients - Providing surgical prophylaxis to prevent postoperative infections in various procedures
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Mac Heal Laboratories Ltd, Gujarat
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Ceftazidime Injection USP 1gm Sterile Antibiotic For Severe Infections
Ceftazidime Injection USP 1gm sterile antibiotic treats severe bacterial infections with reliable quality for hospitals, clinics, and healthcare distributors requiring effective Gram-negative coverage.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Mac Heal Laboratories Ltd
Health & Personal CareManufacturer