Apparel & Fashion
Loading subcategories...
View More
Description
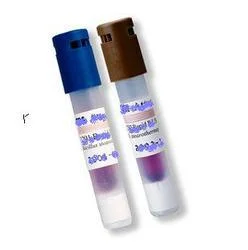
ETO sterilization biological indicators provide critical validation for ethylene oxide sterilization processes, specifically engineered for large-scale medical equipment sterilizat...
ETO sterilization biological indicators provide critical validation for ethylene oxide sterilization processes, specifically engineered for large-scale medical equipment sterilization. These indicators contain highly resistant microorganisms that confirm whether sterilization conditions have been effectively achieved, offering reliable performance for industrial applications. The product ensures that medical devices achieve the sterility assurance levels required by international standards, making them essential for quality control in manufacturing environments where patient safety depends on effective sterilization. Their precise validation capabilities help operations maintain consistent sterilization outcomes across diverse load configurations and cycle parameters. These biological indicators serve numerous industries including medical device manufacturing, pharmaceutical production, hospital sterilization departments, and research laboratories. They are particularly crucial for companies producing disposable medical products such as syringes, catheters, surgical kits, and implantable devices that require ethylene oxide sterilization. The healthcare industry relies on these indicators to maintain compliance with regulatory requirements from organizations like FDA, ISO, and other international health authorities, ensuring that all sterilized products meet strict safety standards before reaching end-users. Their application extends to contract sterilization facilities serving multiple medical device manufacturers. The business value of these biological indicators lies in their reliability and consistency, which directly impacts operational efficiency and risk management. By providing accurate sterilization validation, they help prevent costly product recalls, maintain regulatory compliance, and protect brand reputation. Their consistent performance reduces validation time and costs while ensuring sterilization processes meet required standards. This reliability translates into reduced downtime and increased confidence in sterilization processes, ultimately supporting business continuity and customer trust in the safety of medical products. Their design accommodates various sterilization chamber sizes and load configurations. Key Features: - Specifically designed for ethylene oxide sterilization validation - Suitable for large-scale industrial sterilization cycles - Contains highly resistant biological microorganisms - Consistent and reliable performance across batches - Engineered for precise validation of sterilization parameters Benefits: - Ensures medical device safety through accurate sterilization validation - Supports regulatory compliance with international standards - Reduces risk of product recalls and associated costs - Enhances operational efficiency with reliable performance - Builds customer trust through consistent quality assurance
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
These biological indicators provide essential validation for ethylene oxide sterilization processes in medical device manufacturing and pharmaceutical production. They deliver accurate confirmation that sterilization conditions have been effectively met, ensuring product safety and regulatory compliance for critical healthcare products. Their reliability makes them indispensable for operations processing heat-sensitive medical equipment where alternative sterilization methods are unsuitable. They help maintain stringent quality control standards in environments where patient safety depends on effective sterilization validation and documentation. Real-world applications: - Validating sterilization cycles for disposable syringes, needles, and catheters - Quality assurance testing for surgical instrument manufacturers - Sterilization verification in pharmaceutical production facilities - Monitoring ETO processes for medical device packaging operations - Compliance testing for hospital central sterile supply departments
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Shivaah Packaging, West Bengal
ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
ETO Sterilization Biological Indicators For Medical Device Safety
ETO sterilization biological indicators ensure medical device safety with precise validation for large-scale industrial sterilization cycles. These reliable indicators confirm ethylene oxide sterilization effectiveness for critical medical equipment.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Shivaah Packaging
Manufacturer