Apparel & Fashion
Loading subcategories...
View More
Description
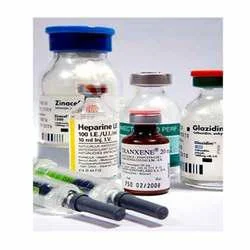
Our premium anticancer lyophilized injectables represent a critical advancement in oncology medication delivery, manufactured through sophisticated freeze-drying technology that re...
Our premium anticancer lyophilized injectables represent a critical advancement in oncology medication delivery, manufactured through sophisticated freeze-drying technology that removes water from liquid formulations under controlled vacuum conditions. This lyophilization process ensures exceptional product stability, significantly extends shelf life, and preserves full pharmacological potency without requiring cold chain storage. We offer a comprehensive portfolio including key agents like Bortezomib, Pemetrexed, Gemcitabine, Bleomycin, and Dacarbazine, each produced under rigorous pharmaceutical manufacturing standards. Every batch undergoes stringent quality control testing to guarantee purity, safety, and therapeutic efficacy, providing medical professionals with injectables they can trust for patient care. These lyophilized injectables are indispensable within the global healthcare and pharmaceutical industries, specifically serving hospitals, specialized cancer treatment centers, oncology clinics, and medical distributors. They form the backbone of numerous chemotherapy protocols for treating various malignancies including multiple myeloma, non-small cell lung cancer, pancreatic cancer, and leukemias. Medical distributors and wholesalers consistently choose our products for their unwavering quality, batch-to-batch consistency, and full compliance with international regulatory standards, enabling them to supply reliable anticancer therapies to healthcare providers across diverse markets. The formulations are expertly designed for straightforward reconstitution, facilitating efficient and accurate preparation in busy clinical environments where time and precision are paramount. The business value of our anticancer lyophilized injectables is substantial, primarily through the elimination of complex cold chain logistics that plague many injectable medications. Their inherent stability dramatically reduces transportation and storage costs while minimizing product waste due to temperature excursions. This reliability ensures product integrity from manufacturing through to point-of-use, giving partners confidence in supply chain continuity. We emphasize robust quality assurance systems, dependable supply timelines, and competitive pricing structures, making our portfolio a strategically sound choice for partners seeking dependable, high-quality anticancer solutions that support both clinical excellence and operational efficiency. Key Features: - Advanced lyophilized formulation ensuring superior stability and extended shelf life without refrigeration. - Comprehensive anticancer portfolio including Bortezomib, Pemetrexed, Gemcitabine, and other key agents. - Manufactured under strict cGMP and international pharmacopeial quality control standards. - Consistent batch-to-batch potency and purity verified through rigorous analytical testing. - Designed for easy and rapid reconstitution with appropriate diluents in clinical settings. Benefits: - Enhanced supply chain reliability through temperature-stable formulations that simplify logistics and storage. - Reduced medication waste and associated costs due to superior product stability and longer shelf life. - Trusted therapeutic consistency and efficacy for healthcare providers administering critical chemotherapy. - Compliance with global quality standards, ensuring products meet stringent safety and regulatory requirements. - Supports efficient clinical workflows with ready-to-use formulations that save preparation time for medical staff.
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Our premium anticancer lyophilized injectables are specifically engineered for oncology treatment protocols, providing healthcare professionals with stable, ready-to-use formulations that maintain potency without refrigeration. These products are essential for chemotherapy administration in settings where precision, reliability, and consistent therapeutic outcomes are non-negotiable, supporting a wide range of cancer treatment regimens from first-line to palliative care. These injectables serve critical roles across multiple medical specialties, including medical oncology, hematology, and clinical research. They are designed for seamless integration into hospital pharmacies, outpatient cancer centers, and specialized treatment facilities, ensuring healthcare providers have access to high-quality anticancer medications that meet stringent international pharmacopeial standards for safety and efficacy. - Administered in hospital oncology departments for precise chemotherapy treatment cycles targeting cancers like multiple myeloma and lung cancer. - Utilized in outpatient chemotherapy clinics for regular infusion therapies, offering stable formulations that simplify storage and handling. - Supplied to hospital and compounding pharmacies for accurate preparation and dispensing of critical anticancer medications. - Employed in clinical research trials and oncology studies investigating new therapeutic protocols and combination treatments. - Distributed to healthcare networks and facilities in regions with challenging logistics, leveraging the stability of lyophilized products for reliable supply.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Adley Formulations, Punjab
ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Main Markets
South Asia
Premium Anticancer Lyophilized Injectables For Hospitals And Clinics
Premium anticancer lyophilized injectables including Bortezomib, Pemetrexed, and Gemcitabine. High-purity, stable formulations for hospital and clinic chemotherapy, trusted by medical distributors worldwide for reliability.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Adley Formulations
Manufacturer