Apparel & Fashion
Loading subcategories...
View More
Description
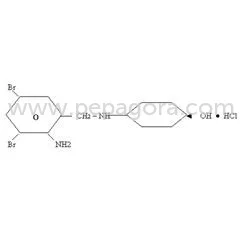
Ambroxol HCL represents a premium-grade active pharmaceutical ingredient specifically engineered for respiratory drug formulations, featuring a guaranteed purity range of 99.0% to ...
Ambroxol HCL represents a premium-grade active pharmaceutical ingredient specifically engineered for respiratory drug formulations, featuring a guaranteed purity range of 99.0% to 101.0% that ensures exceptional consistency in pharmaceutical manufacturing. This white, crystalline powder with molecular formula C13H18Br2N2O·HCl and molecular weight of 414.6 g/mol delivers reliable mucolytic and expectorant properties essential for effective respiratory therapeutics. The compound demonstrates optimal solubility profiles in key pharmaceutical solvents including N,N-dimethylformamide and methanol, while maintaining stability through proper light-sensitive storage conditions. This high-purity API meets rigorous quality standards, providing manufacturers with a trusted foundation for developing respiratory medications that deliver predictable clinical outcomes and patient benefits. The pharmaceutical industry extensively incorporates Ambroxol HCL into respiratory treatment regimens across multiple therapeutic categories, particularly in medications addressing chronic bronchitis, asthma, COPD, and other respiratory disorders. Manufacturers leverage this API's ability to effectively reduce mucus viscosity and enhance pulmonary clearance in various dosage forms including syrups, tablets, capsules, and inhalable solutions. The ingredient's well-established safety profile supports its use across diverse patient demographics, from pediatric formulations to geriatric medications, while its compatibility with numerous excipients enables flexible formulation strategies. Global regulatory compliance ensures seamless integration into manufacturing processes targeting international markets, with documentation supporting pharmacopeial standards across major regulatory regions. Businesses selecting Ambroxol HCL API benefit from unparalleled reliability and batch-to-batch consistency that minimizes production variability and quality control challenges. This consistency translates to reduced manufacturing risks, faster time-to-market for new products, and enhanced end-product performance that meets evolving patient needs. The API's adherence to international quality standards provides assurance of regulatory compliance, while its chemical stability supports extended shelf life and manufacturing flexibility. By incorporating this high-performance ingredient, pharmaceutical manufacturers gain competitive advantage through superior product efficacy, streamlined production processes, and strengthened market positioning with respiratory therapeutics that demonstrate proven clinical effectiveness. Key Features: - Guaranteed purity range between 99.0% and 101.0% - Molecular structure C13H18Br2N2O·HCl with 414.6 g/mol weight - White crystalline powder with odorless characteristics - Optimal solubility in N,N-dimethylformamide and methanol - Light-protected storage requirements for maximum stability Benefits: - Consistent batch-to-batch performance in drug formulations - Enhanced mucus clearance and bronchial relief properties - Flexible compatibility with various pharmaceutical excipients - Reliable stability profile supporting extended product shelf life - Comprehensive documentation for global regulatory compliance
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Ambroxol HCL API serves as a fundamental component in respiratory medication development, offering potent mucolytic action that effectively breaks down thick mucus and improves bronchial clearance. Its high purity profile ensures consistent performance across various pharmaceutical formulations while meeting stringent international pharmacopeial standards for safety and efficacy in respiratory care products. This versatile API demonstrates excellent compatibility with multiple excipients, allowing manufacturers to develop both solid and liquid dosage forms with predictable dissolution characteristics and stable shelf-life performance. Pharmaceutical companies worldwide utilize Ambroxol HCL in developing specialized respiratory treatments targeting chronic conditions including bronchitis, asthma, and COPD. The ingredient's proven safety profile makes it suitable for diverse patient populations from pediatric to geriatric applications. Manufacturers appreciate its formulation flexibility for creating both prescription medications and over-the-counter remedies, supported by comprehensive documentation and quality assurance protocols that streamline regulatory compliance across global markets. - Manufacturing cough syrups and expectorant liquids for effective mucus clearance - Producing fast-dissolving tablets and lozenges for convenient respiratory relief - Developing inhalable solutions and nebulizer treatments for asthma management - Formulating pediatric and adult medications for bronchitis and COPD care - Creating combination therapies with antitussives and anti-inflammatory compounds
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Ambroxol HCL API 99 101% Purity For Respiratory Drugs
Pharmaceutical Ambroxol HCL API with 99-101% purity for respiratory drug manufacturing. This high-quality active ingredient delivers reliable mucolytic and expectorant properties for effective respiratory treatments.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer