Apparel & Fashion
Loading subcategories...
View More
Description
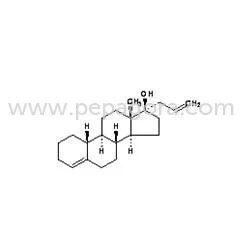
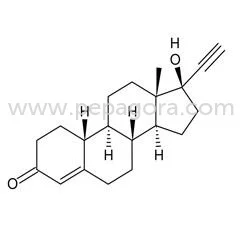
Allylestrenol represents a premium-grade progestogen hormone analog supplied as white to off-white crystalline powder with characteristic odor. Specifically engineered for pharmace...
Allylestrenol represents a premium-grade progestogen hormone analog supplied as white to off-white crystalline powder with characteristic odor. Specifically engineered for pharmaceutical applications, this high-purity compound ensures exceptional stability and compliance with rigorous industry standards. It demonstrates solubility in methanol and chloroform while remaining insoluble in water, undergoing comprehensive testing for identity, potency, and impurities to meet the exacting requirements of pharmaceutical manufacturers and distributors seeking dependable raw materials. This product finds essential application across pharmaceutical manufacturing, biotechnology development, and veterinary medicine sectors. It serves as a fundamental component in producing hormonal therapies, fertility treatments, and gynecological medications. Companies operating in these specialized fields value Allylestrenol for its unwavering quality consistency, which directly supports the creation of safe and effective medications for both human and animal health applications while maintaining regulatory compliance. Businesses benefit from Allylestrenol's proven reliability and strict adherence to quality control protocols, significantly reducing production risks while ensuring end-product safety. Its compatibility with diverse pharmaceutical formulations and consistent compliance with international regulatory standards makes it a preferred choice for serious buyers focused on maintaining quality assurance and operational efficiency throughout their supply chain operations. Key Features: - High-purity white to off-white crystalline powder with characteristic odor - Soluble in methanol and chloroform while insoluble in water - Controlled melting range between 78-81C with moisture content below 1.0% - Purity assay maintained between 97% to 103% with minimal impurities - Clear solution formation in methanol at 65C without insoluble particles Benefits: - Consistent quality ensures reliable therapeutic performance in final products - High purity levels support precise dosing in pharmaceutical formulations - Stable chemical properties enable long shelf life and storage reliability - Compliance with industry standards reduces regulatory compliance risks - Versatile solubility characteristics facilitate diverse manufacturing processes
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Allylestrenol serves as a critical progestogenic hormone in pharmaceutical manufacturing, providing hormonal stability and therapeutic efficacy. Its high purity and consistent performance support precise dosing in medical applications where reliability directly impacts patient outcomes and treatment safety. This compound is essential for developing hormone therapies, fertility medications, and gynecological treatments. Pharmaceutical companies, research institutions, and clinical suppliers rely on its quality to produce effective end-products that meet regulatory standards and address diverse healthcare needs globally. - Formulating oral tablets for hormone therapy and menstrual disorder treatments - Developing fertility medications to support pregnancy and prevent miscarriage - Manufacturing veterinary pharmaceuticals for animal reproductive health management - Serving as intermediate in synthesis of advanced steroid-based drugs - Supporting clinical research and drug development for progestogen studies
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Pr Pharma Source Pvt Ltd, Maharashtra
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Allylestrenol Powder High Purity Progestogenic Hormone
Pharmaceutical Allylestrenol Powder offers high-purity progestogenic hormone for therapeutic formulations, ensuring reliable quality and strict compliance for medical manufacturers and distributors.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Pr Pharma Source Pvt Ltd
Health & Personal CareManufacturer