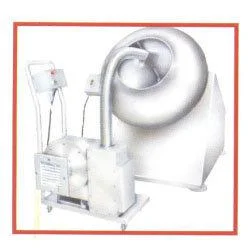
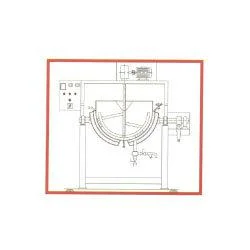
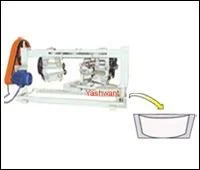
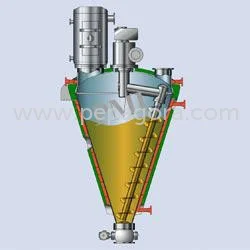
Pharma Grade Mixers (GMP)
24 products available
What are Pharma Grade Mixers (GMP)?
Pharma Grade Mixers (GMP) are specialized equipment designed to ensure the consistent and uniform mixing of pharmaceutical ingredients. These mixers are manufactured in accordance with Good Manufacturing Practices (GMP), which are guidelines set by regulatory authorities to ensure the quality and safety of pharmaceutical products. The design and construction of these mixers focus on hygiene, precision, and efficiency, making them essential in the pharmaceutical industry.
These mixers are primarily used in the production of various pharmaceutical formulations, including powders, granules, and liquid products. Their applications extend to the compounding of medications, blending of active pharmaceutical ingredients (APIs) with excipients, and the preparation of formulations for clinical trials. The pharmaceutical industry heavily relies on these mixers to meet stringent quality standards and regulatory requirements, ensuring that products are safe for consumption.
With the growing demand for pharmaceuticals globally, the market for Pharma Grade Mixers is expanding. Companies are increasingly investing in advanced mixing technologies to enhance product quality and streamline production processes. This trend underscores the importance of reliable and efficient mixing solutions in the competitive landscape of the pharmaceutical sector.
𝗞𝗲𝘆 𝗙𝗲𝗮𝘁𝘂𝗿𝗲𝘀 𝗮𝗻𝗱 𝗦𝗽𝗲𝗰𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharma Grade Mixers (GMP) are characterized by their advanced features that meet the stringent standards of the pharmaceutical industry. These mixers are designed to provide high performance while ensuring compliance with various regulatory requirements.
Key specifications include:
1. Construction Material
- Typically made from stainless steel or other non-reactive materials
- Ensures durability and resistance to contamination
2. Mixing Capacity
- Varies based on model, ranging from small batches to large-scale production
- Capable of handling different volumes of raw material
3. Mixing Mechanism
- Incorporates various technologies such as ribbon blades, paddle mixers, or high-shear mixers
- Provides efficient blending of diverse product formulations
4. Control Systems
- Equipped with advanced digital controls for precise mixing parameters
- Facilitates monitoring of speed, time, and temperature
5. Cleanability
- Designed for easy disassembly and cleaning
- Meets hygiene standards to prevent cross-contamination
6. Compliance with Standards
- Adheres to industry certifications such as CE
- Meets Good Manufacturing Practices (GMP) requirements
7. Customization Options
- Available in various designs to suit specific production needs
- Can be tailored to accommodate unique mixing requirements
8. Safety Features
- Includes safeguards such as overload protection and emergency shut-off
- Ensures operator safety during operation
These specifications highlight the importance of selecting a Pharma Grade Mixer that aligns with production needs and regulatory demands.
𝗖𝗼𝗺𝗺𝗼𝗻 𝗔𝗽𝗽𝗹𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀 𝗮𝗻𝗱 𝗨𝘀𝗲 𝗖𝗮𝘀𝗲𝘀
Pharma Grade Mixers (GMP) find extensive applications across multiple sectors within the pharmaceutical industry. Their versatility allows them to cater to various production requirements.
1. Pharmaceutical Manufacturing: Used for mixing APIs with excipients to create uniform formulations for tablets, capsules, and syrups.
2. Biotechnology: Essential in the preparation of biologics and biopharmaceuticals, where precise blending is crucial for product efficacy.
3. Nutraceuticals: Employed in the formulation of dietary supplements, ensuring even distribution of active ingredients.
4. Veterinary Medicine: Utilized in the production of veterinary drugs and supplements, requiring stringent quality control.
5. Cosmetics: Applied in the formulation of cosmetic products, where consistent texture and quality are vital.
6. Food and Beverage: Used in pharmaceutical food products that require adherence to safety and quality standards.
7. Research and Development: Ideal for small-scale mixing in R&D laboratories, facilitating the development of new drug formulations.
These applications showcase the critical role of Pharma Grade Mixers in ensuring product quality and compliance across various industries.
𝗣𝗿𝗼𝗱𝘂𝗰𝘁 𝗩𝗮𝗿𝗶𝗮𝗻𝘁𝘀 𝗮𝗻𝗱 𝗦𝘂𝗯𝗰𝗮𝘁𝗲𝗴𝗼𝗿𝗶𝗲𝘀
Pharma Grade Mixers (GMP) come in a variety of configurations to meet diverse mixing needs. Each variant offers unique features tailored for specific applications.
High-Shear Mixers
These mixers are designed for applications requiring intense mixing and dispersion. They are ideal for producing emulsions and suspensions, ensuring uniformity in high-viscosity products.
Ribbon Mixers
Ribbon mixers provide effective blending for powders and granules. Their design allows for thorough mixing while minimizing the risk of product degradation, making them suitable for a wide range of pharmaceutical applications.
Batch Mixers
Batch mixers are employed for specific production runs, allowing for flexibility in manufacturing processes. They are commonly used in small to medium-scale operations, providing efficient mixing capabilities.
Continuous Mixers
These mixers are designed for large-scale production, offering streamlined processes for ongoing mixing requirements. They are effective for high-volume applications while maintaining product consistency.
𝗕𝗲𝗻𝗲𝗳𝗶𝘁𝘀 𝗮𝗻𝗱 𝗔𝗱𝘃𝗮𝗻𝘁𝗮𝗴𝗲𝘀
Pharma Grade Mixers (GMP) provide numerous benefits that enhance production efficiency and product quality in the pharmaceutical industry.
1. Enhanced Product Quality: Ensures uniform mixing of ingredients, leading to consistent product quality and efficacy.
2. Compliance Assurance: Meets stringent regulatory standards, helping manufacturers avoid compliance issues.
3. Increased Efficiency: Streamlined mixing processes reduce production times and improve operational efficiency.
4. Versatility: Capable of handling various formulations and batch sizes, accommodating diverse production needs.
5. Improved Safety: Designed with safety features to protect operators and minimize risks during operation.
6. Cost-Effectiveness: Optimized mixing solutions can reduce waste and lower overall production costs.
These advantages make Pharma Grade Mixers a valuable investment for businesses seeking to enhance their manufacturing capabilities.
𝗕𝘂𝘆𝗶𝗻𝗴 𝗚𝘂𝗶𝗱𝗲 𝗳𝗼𝗿 𝗕𝟮𝗕 𝗕𝘂𝘆𝗲𝗿𝘀
When sourcing Pharma Grade Mixers (GMP), it is crucial for B2B buyers to consider various factors to ensure they select the right equipment for their needs.
1. Quality Assessment: Evaluate the quality of mixers by reviewing certifications and compliance with industry standards. Look for mixers that adhere to GMP guidelines.
2. Supplier Verification: Vet potential suppliers to ensure they have a proven track record of providing reliable and efficient mixers. Checking customer reviews and references can be beneficial.
3. Pricing and MOQ: Understand the pricing structure and minimum order quantity (MOQ) requirements. Compare costs across suppliers to make informed purchasing decisions.
4. Delivery and Lead Times: Inquire about logistics and delivery schedules to ensure timely procurement of mixers, especially for production deadlines.
5. Compliance Requirements: Ensure that the mixers meet all regulatory and compliance requirements specific to the pharmaceutical industry in your region.
This guide will help buyers make informed choices when investing in Pharma Grade Mixers.
𝗖𝗼𝗺𝗽𝗹𝗶𝗮𝗻𝗰𝗲 𝗮𝗻𝗱 𝗖𝗲𝗿𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻𝘀
Pharma Grade Mixers (GMP) are subject to rigorous compliance and certification standards to ensure product quality and safety. Key quality standards include ISO 9001, which focuses on maintaining quality management systems, and HACCP, which emphasizes food safety and risk management.
In addition, mixers must comply with Good Distribution Practices (GDP) to ensure the integrity of pharmaceutical products throughout the supply chain. Certifications such as CE indicate that the products meet European safety and health requirements.
Safety testing is a critical component of the manufacturing process, and mixers should undergo thorough evaluations to ensure they meet industry safety standards. Furthermore, regional compliance considerations may vary, requiring manufacturers to stay informed about local regulations.
Adhering to these compliance and certification requirements is essential for companies looking to maintain their reputation and ensure the safety of their products.
Why Source Pharma Grade Mixers (GMP) from Pepagora?
Pepagora offers a reliable platform for sourcing Pharma Grade Mixers (GMP) with several key advantages that cater to the needs of B2B buyers.
Verified Supplier Network: Pepagora connects buyers with a network of verified suppliers, ensuring that all listed manufacturers meet high standards of quality and compliance.
Streamlined RFQ Process: The platform features a straightforward request for quotation (RFQ) system that allows buyers to easily compare offers from multiple suppliers, saving time and effort in the sourcing process.
Trade Assurance and Support: Pepagora provides trade assurance and support to buyers, helping to protect their investments and ensure that transactions are secure and reliable.
Regional Coverage: With a focus on geographic regions such as India, the GCC, and Southeast Asia, Pepagora ensures that buyers can access a diverse range of suppliers and products tailored to their specific needs.
These advantages make Pepagora an ideal choice for businesses looking to source high-quality Pharma Grade Mixers efficiently and effectively.
Newly Added
Recently Added
A.K.Engineering Company
Automatic Paper Core Cutting Machine 25 300mm Inner Diameter 10 1000mm Length
Industrial Paste Preparation Machine Corrosion Resistant 50 300kg
Parallel Paper Tube Making Machine 8 25mm ID 350mm Length Low Maintenance
Asoka Enterprises
Industrial Colloid Mill For High Shear Grinding And Emulsification
Zero Hold Up Filter Press For Chemical Pharmaceutical Food Processing
Automatic Volumetric Liquid Bottle Filling Machine For Pharmaceuticals
Vihar Engineering
Industrial Cream Filling Machine For Cosmetics Pharmaceuticals
Perfume Filling Machine For Glass Plastic Metal Containers
Aluminium Tube Filling Machine For Pharmaceutical Cosmetic Industrial
Yashwanth Industries
Industrial Stainless Steel Heavy Fabrications Durable Corrosion Resistant
Industrial Heat Exchanger Erection Services | High Pressure Systems
Industrial Steel Equipment Fabrication For Construction And Manufacturing
Vihar Engineering
Perfume Filling Machines High Precision Fast Accurate
Pharmaceutical Cosmetic Laminated Tube Filling Machine High Speed 40 50 TPM
Vacuum Filling Machine For Perfumes Pharmaceuticals Cosmetics
Mammon Furnace & Fabricatiors Co
Industrial Cupola Furnace For Foundries Melting Iron Alloys Efficient Durable
Ladle Furnace For Precise Temperature Control In Molten Steel Processing
Industrial Paper Board Mill Machine For High Volume Production
Olympus Engineering Company
Industrial Fluid Bed Dryer For Pharmaceuticals Chemicals Food Processing
Pharmaceutical Hot Air Sterilizer 500L To 2300L Cgmp Compliant
Industrial Octagonal Ribbon Blenders For Dry Powder Mixing
Pm Industries
Industrial Cone Screw Mixers For Powders Slurries And Pastes
Industrial Plough Shear Mixers For Powder Granule Liquid Blending
Industrial Continuous Fluid Bed Dryers For Bulk Material Drying
Pharmac Technologies Pvt Ltd
Industrial Solvent Extraction Unit 25 Liters For Food Pharma
Herbal Extraction Plants With Integrated Lab For Pharma Nutraceutical Cosmetic
Industrial Essential Oil Extraction Systems For Pharmaceutical Perfumery Herbal Food
Yashwant Industries
Industrial Precision Circle Cutting Machine For Mild Steel Sheets
Industrial Metal Fabrication YASHWANT Hand Motorized Out Cutting Machine
Industrial Aluminum Circle Cutter Machine with Adjustable Gauges
Why Businesses Trust Pepagora
Thousands of companies trust Pepagora to source smarter and sell faster
Expand Your Sales Globally
25000+Products listed.Get discovered by global buyers
Tools to boost visibility
Build your own catalog

Source Smarter,Grow Faster
4000+Active RFQs
100+ Countries served
Easy Sourcing Tools