Description
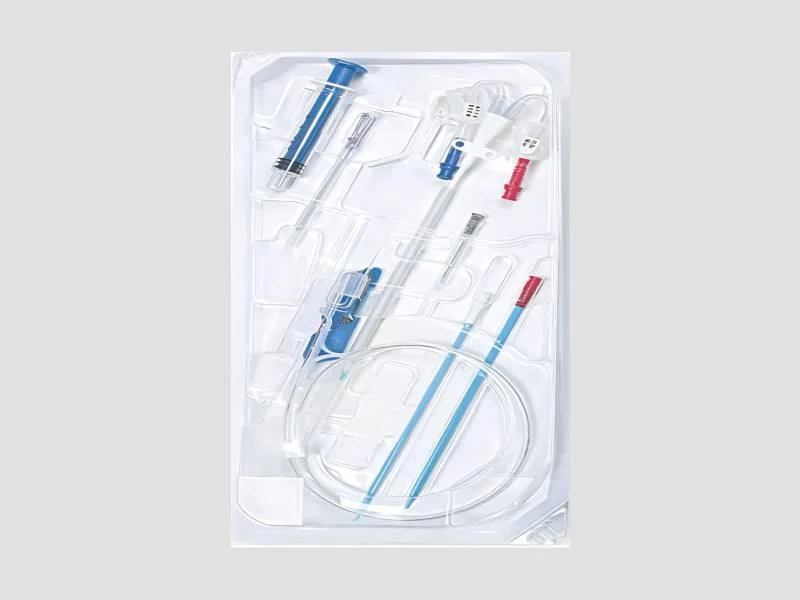
The Plastic Peritoneal Dialysis Catheter Kit represents a complete, sterile, and ready-to-use solution for establishing peritoneal access, a critical procedure in renal replacement...
The Plastic Peritoneal Dialysis Catheter Kit represents a complete, sterile, and ready-to-use solution for establishing peritoneal access, a critical procedure in renal replacement therapy. This kit features a 6 Fr tunneled catheter constructed from medical-grade plastic, designed for controlled insertion with minimal tissue trauma. The tunneled configuration, where the catheter passes under the skin before entering the peritoneal cavity, is a key design element that enhances mechanical stability and creates a biological barrier against infection. The plastic material offers an optimal balance of flexibility and durability, promoting patient comfort during long-term implantation while ensuring reliable performance during fluid exchange. Supplied pre-sterilized, the kit eliminates the need for on-site reprocessing, allowing for immediate use in the operating room or procedure suite, thereby streamlining clinical workflows and supporting strict aseptic technique. This catheter kit is a fundamental tool within nephrology and renal care units, hospitals, and specialized dialysis centers worldwide. It is specifically utilized for patients diagnosed with end-stage renal disease (ESRD) who are candidates for peritoneal dialysis. Medical professionals rely on it to initiate PD therapy, whether for Continuous Ambulatory Peritoneal Dialysis (CAPD) or Automated Peritoneal Dialysis (APD). Beyond initial placement, the kit is also essential for scheduled catheter replacements or revisions necessitated by exit-site infections, tunnel infections, catheter malfunction, or mechanical failure. Its design supports the growing preference for home-based dialysis programs by providing a dependable access point that patients can manage with proper training, contributing to more flexible and patient-centered care models. For healthcare procurement specialists, the value of this kit lies in its combination of clinical efficacy, procedural efficiency, and supply chain reliability. The pre-sterilized, single-use format guarantees product integrity and eliminates cross-contamination risks, directly supporting patient safety protocols. The standardized 6 Fr size and consistent plastic construction ensure predictable performance and handling, reducing the learning curve for surgical teams and minimizing procedural variables. Bulk availability with a minimum order quantity caters to the high-volume needs of institutional buyers, ensuring consistent stock for routine and emergency procedures. This reliability translates into fewer supply disruptions, predictable budgeting for renal care programs, and the assurance of using a device designed for long-term, trouble-free function in a demanding clinical environment. Key Features: - Tunneled catheter design for enhanced subcutaneous stability and reduced infection risk. - Constructed from medical-grade plastic for optimal flexibility, durability, and patient comfort. - Standard 6 Fr size ensuring controlled, minimally traumatic peritoneal insertion. - Supplied as a complete, pre-sterilized kit for immediate use in sterile fields. - Designed specifically for reliable, long-term peritoneal dialysis fluid inflow and outflow. Benefits: - Enables safe and efficient establishment of permanent access for peritoneal dialysis therapy. - Supports both Continuous Ambulatory Peritoneal Dialysis (CAPD) and Automated Peritoneal Dialysis (APD) modalities. - Reduces procedural setup time and complexity with ready-to-use, sterile packaging. - Promotes long-term catheter retention and function through stable tunneled implantation. - Provides a consistent, reliable solution for high-volume clinical and dialysis center procurement.
Specifications
| Attribute | Value |
|---|---|
| Product NameUsage/ApplicationTypeSterilizationMaterialSizeKit Type |
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
This Plastic Peritoneal Dialysis Catheter Kit is engineered to establish permanent, safe access to the peritoneal cavity for patients undergoing dialysis treatment. Its primary function is to facilitate the infusion and drainage of dialysis fluid, enabling the critical removal of metabolic waste and excess fluid from the body. The tunneled design is specifically intended for long-term therapeutic use, providing enhanced stability and a reduced pathway for potential infections compared to non-tunneled alternatives. The kit is indispensable in clinical settings managing end-stage renal disease (ESRD). It is deployed for initiating new peritoneal dialysis (PD) therapy, managing ongoing continuous ambulatory peritoneal dialysis (CAPD) or automated peritoneal dialysis (APD) regimens, and for necessary catheter replacement procedures. Its reliable performance supports consistent treatment delivery in environments dedicated to renal care and patient management. - Establishing initial peritoneal access for patients beginning long-term peritoneal dialysis therapy. - Managing routine fluid exchanges in Continuous Ambulatory Peritoneal Dialysis (CAPD) for chronic kidney failure. - Facilitating Automated Peritoneal Dialysis (APD) cycles during overnight treatments in home or clinical settings. - Replacing existing catheters due to malfunction, infection, or damage in follow-up surgical procedures. - Providing a stable, long-term vascular access solution for patients unsuitable for hemodialysis.
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Medify Surgicals, Tamil Nadu
Health & Personal Care•Distributor / Wholesaler
Factory Details
Factory SizeBelow 1000 sqm
Medical Plastic Peritoneal Dialysis Catheter Kit 6Fr Sterile Tunneled
The Plastic Peritoneal Dialysis Catheter Kit (6 Fr) is a sterilized, tunneled clinical kit for safe peritoneal access. This lightweight plastic catheter ensures smooth insertion and reliable fluid exchange, supplied sterile for immediate procedure readiness.
Min. Order Quantity: 1
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Medify Surgicals
Health & Personal CareDistributor / Wholesaler