Description
Nimodipine BP/USP represents a high-purity calcium channel blocker specifically engineered for cerebral vasodilation applications in pharmaceutical manufacturing. This yellow cryst...
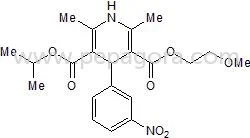
Nimodipine BP/USP represents a high-purity calcium channel blocker specifically engineered for cerebral vasodilation applications in pharmaceutical manufacturing. This yellow crystalline powder, chemically designated as 1,4-Dihydro-2,6-dimethyl-4-(3-nitrophenyl)-3,5-Pyridinedicarboxylic acid 2-methoxyethyl-1-methylethyl ester, demonstrates exceptional solubility in alcohol, chloroform, and ethyl acetate while remaining insoluble in water. With a consistent melting point of 123-126°C, our pharmaceutical-grade Nimodipine ensures reliable performance throughout formulation processes. Manufactured under stringent quality controls, this API delivers predictable therapeutic effects for critical neurological interventions where consistency and purity are non-negotiable. The pharmaceutical industry utilizes Nimodipine primarily within neurology and critical care medicine sectors. Hospital pharmacies and compounding facilities incorporate this API into standardized treatment protocols for subarachnoid hemorrhage patients, where timing and efficacy directly impact recovery outcomes. Neurosurgeons and intensivists depend on Nimodipine's selective cerebral vasodilation properties to prevent secondary ischemic injuries following bleeding events. Global pharmaceutical manufacturers trust this BP/USP compliant ingredient for producing neuroprotective medications that meet international regulatory standards across multiple markets, ensuring patient safety and treatment effectiveness. Businesses selecting our Nimodipine benefit from unmatched reliability and supply chain consistency that supports continuous production schedules. Our manufacturing partners receive comprehensive documentation and regulatory support that facilitates smooth market entry across multiple regions. The product's proven stability profile reduces formulation challenges while maintaining therapeutic efficacy throughout shelf life, providing cost-effective manufacturing solutions. Distributors appreciate the consistent quality that builds trust with medical professionals who depend on this critical medication for patient care in emergency situations. Key Features: - BP and USP compliance for global market access - High purity yellow crystalline powder formulation - Melting point consistently between 123-126°C - Soluble in alcohol, chloroform and ethyl acetate - Stable chemical properties for reliable manufacturing Benefits: - Consistent quality for predictable therapeutic outcomes - Global regulatory compliance simplifies market entry - Reliable performance in various dosage formulations - Proven stability reduces manufacturing challenges - Trusted by medical professionals for critical care
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Nimodipine is formulated into oral tablets and capsules for neurological treatment protocols, specifically targeting cerebral arteries to prevent vasospasm after subarachnoid hemorrhage. Its consistent purity and stability make it essential for pharmaceutical manufacturers developing neurocritical care medications that require reliable performance in various dosage forms. This active pharmaceutical ingredient meets both BP and USP standards, ensuring compliance with international regulatory requirements. Medical facilities administer Nimodipine to prevent ischemic deficits following aneurysmal bleeding events, with its selective cerebral vasodilation proving critical in emergency and intensive care settings where patient outcomes depend on rapid intervention. - Prevents cerebral vasospasm following subarachnoid hemorrhage - Reduces ischemic neurological deficits in stroke patients - Used in hospital emergency and neurology departments - Incorporated into neuroprotective treatment protocols - Manufactured into delayed-release oral dosage forms
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Laksh Finechem Pvt Ltd, Gujarat
Health & Personal Care•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Nimodipine BP/USP High Purity Cerebral Vasodilator For Pharmaceutical Manufacturing
High-purity Nimodipine BP/USP cerebral vasodilator for pharmaceutical manufacturing treats subarachnoid hemorrhage complications with trusted reliability and consistent quality for global distributors.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Laksh Finechem Pvt Ltd
Health & Personal CareManufacturer