Description
Our Metformin Active Pharmaceutical Ingredient represents the highest standard in diabetes treatment manufacturing, sourced from certified facilities following international qualit...
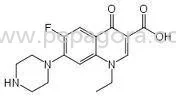
Our Metformin Active Pharmaceutical Ingredient represents the highest standard in diabetes treatment manufacturing, sourced from certified facilities following international quality standards. This high-purity compound ensures exceptional efficacy in glucose regulation through rigorous quality control processes including HPLC testing, heavy metal analysis, and microbial limits testing. With consistent particle size distribution and optimal flow properties, our Metformin enables efficient manufacturing processes for tablet compression and formulation development while maintaining strict compliance with pharmacopeia standards including USP, EP, and IP specifications for global market acceptance and regulatory compliance. Metformin serves as fundamental therapy in diabetes treatment across multiple healthcare sectors and pharmaceutical manufacturing segments. Pharmaceutical companies utilize our API for producing both generic and branded antidiabetic medications serving retail pharmacies, hospital networks, and government health programs. Export-oriented manufacturers choose our Metformin for international markets requiring comprehensive documentation and quality certifications, while healthcare institutions and compounding pharmacies rely on our consistent quality for developing specialized formulations addressing specific patient population needs and treatment protocols in diverse healthcare environments. Businesses selecting our Metformin API gain significant advantages through reliable supply chain partnerships and consistent product quality that reduces production variances and manufacturing challenges. The product's excellent stability profile extends shelf life and minimizes waste in manufacturing operations, while flexible quantity options accommodate everything from research and development projects to large-scale production requirements. Comprehensive documentation including Certificate of Analysis, Stability Data, and Regulatory Support ensures seamless regulatory approvals across multiple markets, with competitive pricing structures that maintain cost-effective manufacturing without compromising the premium quality standards essential for pharmaceutical applications and patient safety. Key Features: - Pharmaceutical-grade purity exceeding 99.5% confirmed by HPLC analysis - Manufactured in WHO-GMP and ISO certified facilities ensuring quality - Consistent particle size distribution for optimal manufacturing processes - Comprehensive documentation package including COA and stability data - Multiple packaging options available from kilogram to ton quantities Benefits: - High purity standards ensure therapeutic efficacy and patient safety - Certified manufacturing facilities guarantee consistent quality and reliability - Optimal physical properties enable smooth production and formulation - Complete documentation supports regulatory compliance and market approval - Flexible packaging solutions meet diverse production scale requirements
Specifications
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Metformin API serves as the primary active ingredient in diabetes treatment formulations worldwide. This pharmaceutical-grade compound effectively manages blood glucose levels by reducing hepatic glucose production and enhancing insulin sensitivity in patients, making it essential for modern diabetes care protocols and medication development programs across healthcare systems. Manufactured under strict Good Manufacturing Practice guidelines, our Metformin API ensures consistent quality and reliable performance in pharmaceutical production. It demonstrates excellent stability and compatibility with common excipients, enabling smooth manufacturing processes for various dosage forms while maintaining therapeutic efficacy and meeting international quality standards for pharmaceutical ingredients. - Production of immediate-release diabetes tablets in 500mg, 850mg, and 1000mg strengths - Manufacturing extended-release formulations for once-daily dosing regimens - Development of combination therapies with other antidiabetic medications - Supply to hospital pharmacies and institutional healthcare providers - Manufacturing export-quality pharmaceutical preparations for international markets
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile
Bavaria Pharma, Gujarat
Raw Materials & Chemicals•ManufacturerDistributor / WholesalerImporter / Exporter
Factory Details
Factory SizeBelow 1000 sqm
Annual Production Capacity0
Pharmaceutical Metformin API For Diabetes Medication Manufacturing
Premium Metformin API for diabetes medication manufacturing offers high-purity active pharmaceutical ingredient with reliable supply for tablets and extended-release formulations, trusted by global pharmaceutical companies.
Min. Order Quantity: 1 units
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Bavaria Pharma
Raw Materials & ChemicalsManufacturer