Description
Chlorpropamide represents a high-purity anti-diabetic active pharmaceutical ingredient manufactured to exacting quality standards, featuring 98.5-101.0% assay concentration and ≤0....
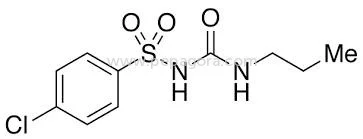
Chlorpropamide represents a high-purity anti-diabetic active pharmaceutical ingredient manufactured to exacting quality standards, featuring 98.5-101.0% assay concentration and ≤0.1% maximum impurities. This sulfonylurea compound, chemically identified as 4-Chloro-N-[(propylamino) carbonyl]-1[(p-chlorophenyl) sulfonyl]-3-propylurea, serves as a fundamental component in diabetes treatment medications. Our OEM supply model guarantees consistent quality across all batches, supported by negotiable pricing structures and reliable on-time delivery to align with your production timelines. Each batch undergoes comprehensive quality control testing to ensure compliance with pharmacopeial standards for pharmaceutical applications. The pharmaceutical manufacturing sector extensively utilizes chlorpropamide in producing oral anti-diabetic medications specifically for type 2 diabetes management. This API holds particular value for pharmaceutical companies specializing in endocrinology products, generic medication manufacturers, and research institutions focused on diabetes treatment advancements. Healthcare providers and medical facilities depend on medications containing this ingredient for implementing effective diabetes management protocols. The compound's well-established therapeutic profile positions it as a preferred choice for formulators creating reliable and effective anti-diabetic formulations for global healthcare markets. Organizations selecting our chlorpropamide benefit from exceptional reliability, unwavering quality consistency, and robust supply chain stability. The product's high purity guarantees optimal formulation performance and regulatory compliance, significantly reducing production risks while ensuring end-product therapeutic efficacy. Our manufacturing approach allows for specific requirement accommodations to support unique formulation needs. The competitive pricing framework and dependable delivery scheduling provide substantial value to businesses seeking to maintain uninterrupted production flows and effectively meet evolving market demands without compromise. Key Features: - High purity pharmaceutical grade with precise 98.5-101.0% assay concentration - Exceptionally low impurity levels guaranteed at ≤0.1% maximum - OEM manufacturing support for specialized requirement accommodation - Rigorous quality control protocols adhering to pharmacopeial standards - Consistent chemical properties ensuring formulation reliability Benefits: - Ensures optimal therapeutic performance in final medication formulations - Reduces production risks through guaranteed purity and quality standards - Supports regulatory compliance and streamlined approval processes - Provides formulation consistency batch after batch for product reliability - Enhances manufacturing efficiency through dependable supply chain partnerships
Specifications
| Attribute | Value |
|---|---|
| Type | Chlorpropamide |
| Impurity | ≤0.1% |
| Ignition | ≤0.1% |
| Supply Type | OEM |
| Price | Negotiable |
| Delivery | On Time Delivery |
| Application | Industrial Applications |
| Assay | 98.5-101.0% |
| Therapeutic Usage | Anti-Diabetic |
| Chemical Name | Benzene Sulfonamide, 4-Chloro-N-[(propylamino) carbonyl] -1[(p-chlorophenyl) sulfonyl]-3-propylurea |
| Additional Information | |
|---|---|
| Country of Origin | India |
| Customisable | No |
Application
Chlorpropamide API is a sulfonylurea-class anti-diabetic active pharmaceutical ingredient specifically designed for type 2 diabetes treatment formulations. It functions by stimulating insulin secretion from pancreatic beta cells while reducing hepatic glucose production, providing effective blood sugar control through consistent therapeutic action in oral medications. This high-purity pharmaceutical compound offers excellent stability and bioavailability, making it ideal for various formulation types. Manufacturers worldwide trust chlorpropamide for its proven efficacy in glucose regulation and consistent performance in meeting international pharmacopeia standards for anti-diabetic drug production. Real-world applications: - Manufacturing oral anti-diabetic tablets for type 2 diabetes management - Producing extended-release formulations for sustained blood glucose control - Developing combination therapies with complementary anti-diabetic agents - Pharmaceutical research and development of innovative diabetes treatments - Compounding specialized medications for individualized treatment protocols
Trade Details
| Available Stock | In stock |
| Sample Availability | No |
Payment Terms
| Payment Terms |
|
Company Profile

Kothari Phyto Chemicals International, Tamil Nadu
Health & Personal Care•ManufacturerImporter / Exporter
Factory Details
Factory Size1000-5000 sqm
Annual Production Capacity0
Main Markets
Eastern Asia
Chlorpropamide API Anti Diabetic Pharmaceutical Ingredient 98.5 101.0% Assay
Chlorpropamide API anti-diabetic pharmaceutical ingredient with 98.5-101.0% assay and ≤0.1% impurities for reliable diabetes medication manufacturing and OEM supply with on-time delivery.
Min. Order Quantity: 1 Tons
Shipping
Shipping fee and delivery date to be negotiated. Contact supplier now for more details.
Kothari Phyto Chemicals International
Health & Personal CareManufacturer